Pre Lab
The reactants phenolphthalein, agarose, and sodium hydroxide were combined to create the party gel. Phenolphthalein was to give the gel a pink color. We cut out an apple shape and a tube shape out of the phenolphthalein. The gel appeared to by soft, but was actually thick and tough. We used a potato corer and an apple shaped cutter to produce two different sections of the gel; each having different surface areas. We placed 10 mL of vinegar in each beaker, that the gel shapes were contained in. We added 10 additional mL of vinegar after three minutes. We added the vinegar to the gel to see how quickly the vinegar would diffuse into the party gel. As we poured the vinegar over the the gel, it became transparent. After about ten minutes, neither of the pieces were completely clear. Since the surface area to volume ratio of the core was higher than the ratio of the apple, the vinegar to diffuse more quickly, and the core became clear faster. Thus, surface area played a part in the reaction.
Lab Materials and Procedure
The materials used were: potato cores, five solutions with different amounts of sucrose (50mL each), five beakers, a petri dish, and a scale.
First, cut the potato into five roughly equal weights. Weigh each core. Place 50 mL of each solution into different beakers. Put each core into different solutions. Weigh each core again after thirty minutes of being place in the solution. Whichever piece has lost the most weight, was submerged in the solution containing the most sugar. The independent varible is what type of solution and the dependent is the weight of the potato.
First, cut the potato into five roughly equal weights. Weigh each core. Place 50 mL of each solution into different beakers. Put each core into different solutions. Weigh each core again after thirty minutes of being place in the solution. Whichever piece has lost the most weight, was submerged in the solution containing the most sugar. The independent varible is what type of solution and the dependent is the weight of the potato.
Hypothesis

If we were to place five potato cores into five different unknown sugar solutions, ranging in different amounts, then solution A will cause the core to loose the most weight, and the solution D will cause the core to loose the least weight. Thus, we predict that solution A will be the most hypertonic and solution D will be the most hypotonic.
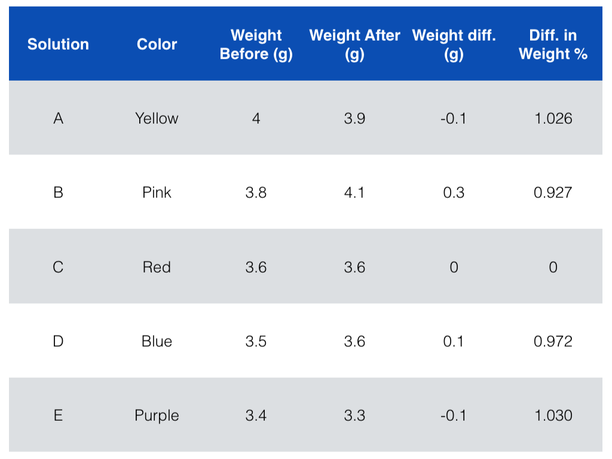
Data Table
Conclusion
The order of solutions from most concentrated to least concentrated is the following: pink, blue, red, yellow, purple.
The solute potential:
A.) (-1)(.8)(.0831)(295)= -19.612
B.) (-1)(.2)(.0831)(295)= -4.903
C.) (-1)(.6)(.0831)(295)= -14.709
D.) (-1)(.4)(.0831)(295)= -9.806
E.) (-1)(1.0)(.0831)(295)= -24.764
If we left them over the weekend, the cells in a hypotonic solution would not burst, because the cell wall would keep it from bursting.
If this experient were to be done with animal cells/ tissue, it would not be effective. The animal cells/ tissue would not be effective because they would burst, unlike the plant cells.
Sources of error were the fact that the time of thirty minutes was not exact for each of the solution. For instance there was a time period between the submersion of the first potato core and the last potato core. This produced error because each core was not left in the solution for the exact same ammount of time. Another source would be that the weights of the potato cores were not exact. The experient would be more accurate if each was the same weight. Also, we spilt one of the beakers. This could have led to error in the calculations.
The solute potential:
A.) (-1)(.8)(.0831)(295)= -19.612
B.) (-1)(.2)(.0831)(295)= -4.903
C.) (-1)(.6)(.0831)(295)= -14.709
D.) (-1)(.4)(.0831)(295)= -9.806
E.) (-1)(1.0)(.0831)(295)= -24.764
If we left them over the weekend, the cells in a hypotonic solution would not burst, because the cell wall would keep it from bursting.
If this experient were to be done with animal cells/ tissue, it would not be effective. The animal cells/ tissue would not be effective because they would burst, unlike the plant cells.
Sources of error were the fact that the time of thirty minutes was not exact for each of the solution. For instance there was a time period between the submersion of the first potato core and the last potato core. This produced error because each core was not left in the solution for the exact same ammount of time. Another source would be that the weights of the potato cores were not exact. The experient would be more accurate if each was the same weight. Also, we spilt one of the beakers. This could have led to error in the calculations.